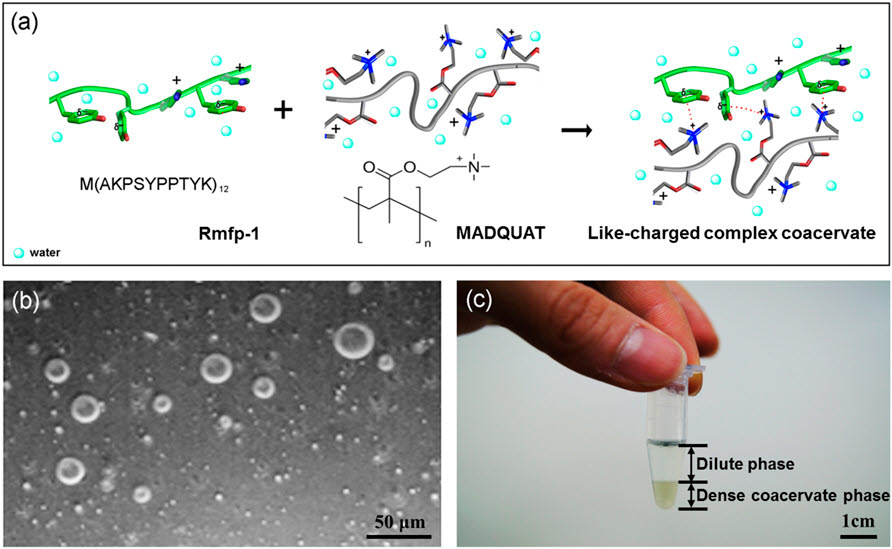
It is well known that polyelectrolyte complexes and coacervates can form on mixing oppositely charged polyelectrolytes in aqueous solutions, due to mainly electrostatic attraction between the
oppositely charged polymers. Here, we report the first (to the best of our knowledge) complexation and coacervation of two positively charged polyelectrolytes, which provides a new paradigm for engineering strong, self-healing interactions between polyelectrolytes underwater and a new marine mussel-inspired underwater adhesion mechanism. Unlike the conventional complex coacervate, the like-charged coacervate is aggregated by strong short-range cation–π interactions by overcoming repulsive electrostatic interactions. The resultant phase of the like-charged coacervate comprises a thin and fragile polyelectrolyte framework and round and regular pores, implying a strong electrostatic correlation among the polyelectrolyte frameworks. The like-charged coacervate possesses a very low interfacial tension, which enables this highly positively
charged coacervate to be applied to capture, carry, or encapsulate anionic biomolecules and particles with a broad range of applications.


 [16.02월] [Angewandte Chemie] 황동수 교수 연구실 Nanomechanics of Poly(catecholamine) Coatings in Aqueous Solutions
[16.02월] [Angewandte Chemie] 황동수 교수 연구실 Nanomechanics of Poly(catecholamine) Coatings in Aqueous Solutions [16.02월] [PNAS] 황동수 교수 연구실 Complexation and coacervation of like-charged polyelectrolytes inspired by mussels
[16.02월] [PNAS] 황동수 교수 연구실 Complexation and coacervation of like-charged polyelectrolytes inspired by mussels
 [15.12월] [PNAS] 국종성 교수 연구실 Amplified Arctic warming by phytoplankton under
[15.12월] [PNAS] 국종성 교수 연구실 Amplified Arctic warming by phytoplankton under